The effectiveness of heparin as an adjunct to fibrinolytic therapy and aspirin in acute MI is uncertain. In patients with large anterior infarctions, moderate-dose heparin (12 500 U SC every 12 hours) reduces the incidence of mural thrombosis detected by 2-dimensional echocardiography by >50%. When used in combination with aspirin in patients with unstable angina or non–Q-wave MI, heparin reduces the short-term rates of cardiovascular death and MI by ≈30% compared with aspirin alone. When used with thrombolytic agents or GP IIb/IIIa antagonists, IV heparin in full doses increases the risk of bleeding, and therefore, the dose should be reduced. It is probably also effective as an adjunct to aspirin and certain thrombolytic agents in patients with evolving MI, and when combined with IV platelet GP IIb/IIIa antagonists in those undergoing high-risk coronary angioplasty. Specifically, IV heparin is effective when combined with aspirin in unstable angina. The totality of evidence supports the view that IV heparin reduces death and MI in patients with acute coronary syndromes. The dose of heparin should be reduced when used concurrently with fibrinolytic agents or IV platelet glycoprotein (GP) IIb/IIIa receptor antagonists. Standardization can be achieved by calibration against plasma heparin concentration by using a therapeutic range of 0.3 to 0.7 U/mL, based on an anti–factor Xa chromogenic assay, or a heparin level of 0.2 to 0.4 U/mL, by protamine sulfate titration. Various heparin-dose-adjustment nomograms have been developed, but none are applicable to all APTT reagents, and the therapeutic range must be tailored accordingly. The APTT should be measured ≈6 hours after a bolus dose of heparin, and the continuous intravenous (IV) dose should be adjusted according to the result. The value of the APTT is limited because commercial APTT reagents vary considerably in responsiveness to heparin. 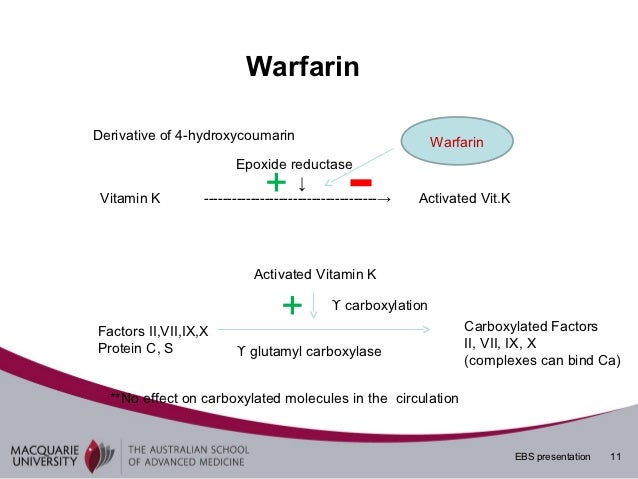
Other limitations include (1) the inability of heparin to inactivate factor Xa in the prothrombinase complex or thrombin bound to fibrin or to subendothelial surfaces and (2) the complications of heparin-induced thrombocytopenia and osteopenia.īecause the anticoagulant response to heparin varies among patients with thromboembolic disorders, it is standard practice to adjust the dose of heparin and monitor its effect by measurement of the activated thromboplastin time (APTT) or, when very high doses are used, by the activated clotting time (ACT). AT-independent binding to macrophages and endothelial cells also results in dose-dependent clearance. Pharmacokinetic limitations are caused by AT-independent binding of heparin to plasma proteins, proteins released from platelets, and endothelial cells, resulting in a variable anticoagulant response and the phenomenon of heparin resistance. The main limitation of heparin results from its propensity to bind to positively charged proteins and surfaces. By inactivating thrombin, heparin not only prevents fibrin formation but also inhibits thrombin-induced activation of platelets and of factors V and VIII. In contrast, very small heparin fragments containing the pentasaccharide sequence inhibit factor Xa via AT. Molecules of heparin with fewer than 18 saccharides lack the chain length to bridge between thrombin and AT and therefore are unable to inhibit thrombin. For inhibition of thrombin, heparin must bind to both the coagulation enzyme and AT, whereas binding to the enzyme is not required for inhibition of factor Xa. Heparin binds to AT through a high-affinity pentasaccharide, which is present on about a third of heparin molecules. It produces its major anticoagulant effect by inactivating thrombin and activated factor X (factor Xa) through an antithrombin (AT)-dependent mechanism. Heparin is a sulfated polysaccharide with a molecular weight range of 3000 to 30 000 Da (mean, 15 000 Da). Mechanism of Action and Pharmacology of Unfractionated Heparin
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
